 |
|
|
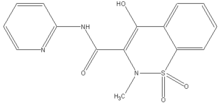
Piroxicam
|
|
| Systematic (IUPAC) name | |
| 9-(hydroxy-pyridin-2-ylamino-methylidene)- 8-methyl-7,7-dioxo-7$l^{6}-thia-8-azabicyclo[ 4.4.0] deca-1,3,5-trien-10-one | |
| Identifiers | |
| CAS number | 36322-90-4 |
| ATC code | M01AC01 M02AA07, S01BC06 |
| PubChem | 5280452 |
| DrugBank | APRD01187 |
| Chemical data | |
| Formula | C15H13N3O4S |
| Mol. weight | 331.348 g/mol |
| Pharmacokinetic data | |
| Metabolism | 4-10% renal |
| Half life | 30 to 86 hours |
| Excretion | 4-10% renal |
| Therapeutic considerations | |
| Pregnancy cat. | C, D if used in the third trimester or near delivery |
Piroxicam (US trade name Feldene) is a nonsteroidal anti-inflammatory drug used to relieve the symptoms of arthritis, primary dysmenorrhoea, pyrexia; and as an analgesic, especially where there is an inflammatory component. It is also used in veterinary medicine to treat certain neoplasias expressing cyclooxygenase (COX) receptors, such as bladder, colon, and prostate cancers.
Mechanism of action
Piroxicam is an NSAID and, as such, is a non-selective COX inhibitor possessing both analgesic and antipyretic properties.
Adverse effects
Piroxicam use can result in gastrointestinal toxicity, tinnitus, dizziness, headache, rash, and pruritus. The most severe adverse reactions are peptic ulceration and gastrointestinal bleeding. Approximately 30% of all patients receiving daily doses of 20 mg of piroxicam experience side effects.[1]
Footnotes
- ^ New Zealand Medicines and Medical Devices Safety Authority. Candyl Medicines datasheet. Retrieved on 2006-09-10.




 216.73.216.16
216.73.216.16 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xx
216.73.xxx.xx
 Server Time:
Server Time:
