| Mescaline | |
|---|---|
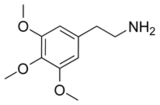
| Chemical name | 3,4,5-Trimethoxy- phenethylamine or 2-(3,4,5-trimethoxyphenyl) ethanamine |
| Chemical formula | C11H17NO3 |
| Molecular mass | 211.26 g/mol |
| Melting point | 128129 °C |
| CAS numbers | 54-04-6 |
| SMILES | NCCC1=CC(OC)=C(OC)C(OC)=C1 |
 |
|
Mescaline (3,4,5-trimethoxyphenethylamine) is a hallucinogenic alkaloid of the phenethylamine class.
It occurs naturally in the peyote cactus (Lophophora williamsii), the San Pedro cactus (Echinopsis pachanoi), in the Peruvian Torch cactus (Echinopsis peruviana), and it is also found in a number of other members of the Cactaceae. It can be extracted from these sources. Mescaline was first isolated and identified in 1897 by the German Arthur Heffter and first synthesized in 1919 by Ernst Späth.
Contents |
Usage and history
The use of extract from peyote in Native American religious ceremonies has been noted since the earliest European contact, notably by the Huichols in Mexico.
Dosage and effects
For the drug to take effect, disk-shaped buttons are cut from the roots, on the top of the cactus, and dried. It is chewed to produce its effect or soaked in water for an intoxicating drink. The effective human dosage is 0.30.5 grams, with the effects lasting for up to 12 hours. Users typically experience visual hallucinations and radically altered states of consciousness, often experienced as pleasurable and illuminating but occasionally as accompanied by feelings of anxiety or revulsion. It is not physically addictive.
Legal status
In the US it was made illegal in 1970 by the Comprehensive Drug Abuse Prevention and Control Act. It was prohibited internationally by the 1971 Convention on Psychotropic Substances[1] and is categorised as a Schedule I hallucinogen by the CSA.
Chemistry
A common synthetical approach starts from 3,4,5-trimethoxybenzaldehyde (PiHKAL entry).
Side effects
One or more of the following side effects may or may not accompany any individual experience with mescaline.
- Open eye visuals
Closed-eye visuals
New thought processes
Dream-like scenarios
Euphoria
Mystical experience
Pupil dilation
Sensations of warm and cold
Temporary splitting/destruction of ego
Dizziness
Vomiting
Tachycardia
Diarrhea
Headaches
Anxiety
Feeling of dying or genocide
Fear of not being able to return to normal consciousness
Hallucinogen persisting perception disorder (HPPD)
Irrationality of the thought-process
Famous users
- Jerry Garcia
Aldous Huxley
Carlos Santana
Ernst Jünger [2]
Leo Kenney [3]
Henri Michaux [4]
Jim Morrison
Carlos Castaneda
Hunter S. Thompson
Allen Ginsberg
Anton Malinsky
Robert Del Naja
Timothy Leary
Christopher Mayhew, Labour MP and BBC television personality, who took Mescaline Hydrochloride in 1955 for an unbroadcast episode of Panorama [5]
Jean-Paul Sartre
Antonin Artaud
Sébastien Branconnier
Comedian Jim Breuer, in his stage act, talks about trying it once as a teenager, in a bit called "The Wizard".
Stanisław Ignacy Witkiewicz, Polish writer, dramatist, photographer, philosopher and painter.
Don Henley, Glenn Frey, Joe Walsh and Don Felder from The Eagles.
Helen Reddy, singer, actress, and feminist activist
Roland Deschain (AKA The Gunslinger), Although a fictional user - Roland's use of Mescaline is prevalent in the first book of the 'Dark Tower' cycle. Roland uses the drug in order to commune with a Demon in a stone circle, and protect the child Jake.
In the film, The Royal Tenenbaums, the character Eli Cash admits to using Mescaline in a nonchalant manner. At the end of the film, it is possible he used Mescaline when he crashed his car into the Tenenbaums' house.
In the film Domino (featuring Keira Knightley, Lucy Liu & Mickey Rourke), the bounty hunters have their coffee spiked with Mescaline as they attempt their escape.
In the novel Fear and Loathing in Las Vegas and its film adaptation, Raoul Duke uses Mescaline.
In the film The Matrix, one of Neo's friends Choi says that he uses Mescaline as it is "the only way to fly". But Yeshwanth disagrees to that statement.
See also
External links
- Mescaline at Erowid
- PiHKAL entry
- Mescaline: The Chemistry and Pharmacology of its Analogs Essay by Alexander Shulgin




 216.73.216.133
216.73.216.133 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xxx
216.73.xxx.xxx
 Server Time:
Server Time: