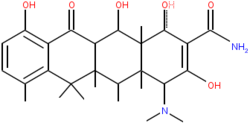
Tetracyclines are a group of broad-spectrum antibiotics whose general usefulness has been reduced with the onset of bacterial resistance. Despite this, they continue to remain the treatment of choice for some specific indications.
They are so named for their four (tetra-) hydrocarbon rings (-cycl-) derivation (-ine).
Contents |
History
The first member of the group to be discovered was Chlortetracycline (Aureomycin) in the late 1940s by Dr. Benjamin Duggar, a scientist employed by Lederle Laboratories who derived the substance from a golden-colored, fungus-like, soil-dwelling bacterium named Streptomyces aureofaciens. Oxytetracycline (Terramycin) was discovered shortly afterwards by AC Finlay et al, it came from a similar soil bacterium named Streptomyces rimosus. Robert Burns Woodward determined the structure of Oxytetracycline enabling Lloyd H. Conover to successfully produce tetracycline itself as a synthetic product. The development of many chemically altered antibiotics formed this group. In June 2005, tigecycline, the first member of a new subgroup of tetracyclines named glycylcyclines was introduced to treat infections which are resistant to other antimicrobics including conventional Tetracyclines.
Examples of tetracyclines

- Naturally-occurring
- Tetracycline
- Chlortetracycline
Oxytetracycline
Demeclocycline
- Semi-synthetic
- Doxycycline
- Lymecycline
Meclocycline
Methacycline
Minocycline
Rolitetracycline
Tigecycline may also be considered a tetracycline antibiotic, though it is usually classified as a glycylcycline antibiotic.
Indication
Tetracyclines may be used in the treatment of infections of the respiratory tract, sinuses, middle ear, urinary tract, intestines, and also gonorrhoea, especially in patients allergic to β-lactams and macrolides; however, their use for these indications is less popular than it once was due to widespread resistance development in the causative organisms.
Their most common current use is in the treatment of moderately severe acne and rosacea (tetracycline, oxytetracycline, doxycycline or minocycline).
Doxycycline is also used as a prophylactic treatment for infection by Bacillus anthracis (anthrax) and is effective against Yersinia pestis, the infectious agent of bubonic plague. It is also used for malaria treatment and prophylaxis, as well as treating elephantiasis.
They remain the treatment of choice for infections caused by chlamydia (trachoma, psittacosis, salpingitis, urethritis and L. venereum infection), Rickettsia (typhus, Rocky Mountain spotted fever), brucellosis, and spirochetal infections (borreliosis, syphilis, and Lyme disease). In addition, they may be used to treat anthrax, plague, tularemia, and Legionnaires disease.
They may have a role in reducing the duration and severity of cholera, although drug-resistance is occurring[1] and their effects on overall mortality is questioned.[2]
Demeclocycline has an additional use in the treatment of SIADH.
Tetracycline derivatives are currently being investigated for the treatment of certain inflammatory disorders.
Administration
When ingested, it is usually recommended that tetracyclines should be taken with a full glass of water two hours after eating, and one hour before eating. This is partly due to the fact that tetracycline binds easily with magnesium, aluminium, iron, and calcium, which reduces its ability to be completely absorbed by the body. Dairy products or preparations containing iron are not recommended directly after taking the drug.
Cautions
Tetracyclines should be used with caution in those with liver impairment and may worsen renal failure (except doxycycline and minocycline). They may increase muscle weakness in myasthenia gravis and exacerbate systemic lupus erythematosus. Antacids and milk reduce the absoption of tetracyclines.
Like many antibiotics, they decrease the effectiveness of birth control pills.
Drugs in the tetracycline class become toxic over time, so expired prescriptions of these drugs should be discarded after the expiration date has passed.
Contraindications
Tetracycline use should be avoided in pregnant or lactating women, and in children with developing teeth because they may result in permanent staining (dark yellow-gray teeth with a darker horizontal band that goes across the top and bottom rows of teeth), and possibly affect the growth, of teeth and bones.
Side effects
Side effects from tetracyclines are not always common, but of particular note is possible photosensitive allergic reaction which increases the risk of sunburn under exposure to UV light from the sun or other sources. This may be of particular importance for those intending to take on holidays long-term doxycyline as a malaria prophylaxis.
They may cause stomach or bowel upsets, and rarely allergic reactions. Very rarely severe headache and vision problems may be signs of dangerous secondary intracranial hypertension also known as Pseudotumor cerebri.
Mechanism and resistance
Tetracycline inhibits cell growth by inhibiting translation. It binds to the 16S part of the 30S ribosomal subunit and prevents the amino-acyl tRNA from binding to the A site of the ribosome. The binding is reversible in nature.
Cells become resistant to tetracycline by at least three mechanisms: enzymatic inactivation of tetracycline, efflux and ribosomal protection. Inactivation is the rarest type of resistance, where an acetyl group is added to the molecule, causing inactivation of the drug. In efflux, a resistance gene encodes a membrane protein that actively pumps tetracycline out of the cell. This is the mechanism of action of the tetracycline resistance gene on the artificial plasmid pBR322. In Ribosomal protection a resistance gene encodes a protein which can have several effects depending on what gene is transferred. Six classes of ribosomal protection genes/proteins have been found, all with high sequence homology suggesting a common evolutionary ancestor. Possible mechanisms of action of these protective proteins include: 1) blocking tetracyclines from binding to the ribosome, 2) binding to the ribosome and distorting the structure to still allow t-RNA binding while tetracycline is bound, and 3) binding to the ribosome and disloding tetracycline. All of these changes to ribosomes are reversible (non-covalent) because ribosomes isolated from both tetracycline resistant and susceptible organisms both bind tetracycline equally well in vitro.
References
- ^ Bhattacharya SK, National Institute of Cholera and Enteric Diseases (2003). "An evaluation of current cholera treatment". Expert Opin Pharmacother 4 (2): 141-6. PMID 12562304.
- ^ Parsi VK (2001). "Cholera". Prim. Care Update Ob Gyns 8 (3): 106-109. PMID 11378428.
- British National Formulary 49 March 2005




 216.73.216.110
216.73.216.110 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xxx
216.73.xxx.xxx
 Server Time:
Server Time:
