| Sulfur dioxide | |
|---|---|
 |
|
| General | |
| Systematic name | sulfur dioxide |
| Other names | Sulfur(IV) oxide Sulfurous anhydride, 'sulphurous anhydride |
| Molecular formula | SO2 |
| Molar mass | 64.054 g mol−1 |
| Appearance | colourless gas |
| CAS number | [7446-09-5] |
| EINECS number | 231-195-2 |
| Properties | |
| Density and phase | 2.551 g/L, gas |
| Solubility in water | 9.4 g/100 mL (25 °C) |
| Melting point | −72.4 °C (200.75 K) |
| Boiling point | −10 °C (263 K) |
| Critical Point | 157.2°C at 7.87 MPa |
| Acidity (pKa) | 1.81 |
| Structure | |
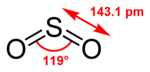
| Molecular shape | bent |
| Dipole moment | 1.63 D |
| Thermodynamic data | |
|
Standard enthalpy of formation ΔfH°gas |
−296.84 kJ mol−1 |
|
Standard molar entropy S°gas |
248.21 J K−1 mol−1 |
| Safety data | |
| EU classification | Toxic |
| R-phrases | R23, R34 |
| S-phrases | S1/2, S9, S26 S36/37/39, S45 |
| NFPA 704 |

0
3
0
|
| PEL-TWA (OSHA) | 5 ppm (13 mg m−3) |
| IDLH (NIOSH) | 100 ppm |
| Flash point | non-flammable |
| RTECS number | WS4550000 |
| Related compounds | |
| Other cations |
Selenium dioxide Tellurium dioxide |
| Related compounds |
Sulfur trioxide Sulfuric acid |
|
Except where noted otherwise, data are given
for materials in their standard state (at 25 °C, 100 kPa) |
|
Sulfur dioxide (also sulphur dioxide) is the chemical compound with the formula SO2. This important gas is the main product from the combustion of sulfur compounds and is of significant environmental concern. SO2 is often described as the "smell of burning sulfur."
SO2 is produced by volcanoes and in various industrial processes. Since coal and petroleum contain various amounts of sulfur compounds, their combustion generates sulfur dioxide. Further oxidation of SO2, usually in the presence of a catalyst such as NO2, forms H2SO4, and thus acid rain.
Contents |
Preparation
Sulfur dioxide can be prepared by burning sulfur:
- S8(s) + 8O2(g) → 8SO2(g)
The combustion of hydrogen sulfide and organosulfur compounds proceeds similarly.
- 2H2S(g) + 3O2(g) → 2H2O(g) + 2SO2(g)
The roasting of sulfide ores such as iron pyrites and sphalerite (zinc blende) also gives SO2:
- 4FeS2(s) + 11O2(g) → 2Fe2O3(s) + 8SO2(g)
- 2ZnS(s) + 3O2(g) → 2ZnO(s) + 2SO2(g)
When anhydrous CaSO4 is heated with coke and sand in the manufacture of cement, CaSiO3, sulfur dioxide is a by-product.
- 2CaSO4(s) + 2SiO2(s) + C(s) → 2CaSiO3(s) + 2SO2(g) + CO2(g)
Action of hot concentrated sulphuric acid on copper tunings will produce sulphur dioxide.
- Cu(s) + 2H2SO4(l) → CuSO4(s) + SO2(g) + 2H2O(l)
Structure and bonding
SO2 is a bent molecule with C2v symmetry point group.
In terms of electron-counting formalisms, the sulfur atom has an oxidation state of +4, a formal charge of 0, and is surrounded by 5 electron pairs. From the perspective of molecular orbital theory, most of these electron pairs are non-bonding in character, as is typical for hypervalent molecules.
Uses
Sulfur dioxide is sometimes used as a preservative in alcoholic drinks, or dried apricots and other dried fruits due to its antimicrobial properties. The preservative is used to maintain the appearance of the fruit rather than prevent rotting. This can give fruit a distinctive chemical taste.
Sulfur dioxide is also a good reductant. In the presence of water, sulfur dioxide is able to decolorize substances that can be reduced by it; thus making it a useful reducing bleach for papers and delicate materials such as clothes.
This bleaching effect normally does not last very long. Oxygen in the atmosphere reoxidizes the reduced dyes, restoring the color. This might explain why older newspapers turn yellow, because paper used for newspaper is naturally yellow.
Sulfur dioxide is also used to make sulfuric acid, being converted to sulfur trioxide, and then to oleum, which is made into sulfuric acid. Sulfur dioxide for this purpose is made when sulfur combines with oxygen. This is called the contact process.
According to Claude Ribbe in The Crime of Napoleon, sulfur dioxide gas was used as an execution poison by the French emperor to suppress a slave revolt in Haiti early in the 19th century.
Prior to the development of Freons, sulfur dioxide was used as a refrigerant in home refrigerators.
H2SO3 is also called "hydrogen sulfite" or sulfurous acid.
Emissions
According to the US EPA (as presented by the 2002 World Almanac or in chart form [1]), the following amount of sulfur dioxide was released in the U.S. per year, measured in thousands of short tons:
| *1999 | 18,867 |
| *1998 | 19,491 |
| *1997 | 19,363 |
| *1996 | 18,859 |
| *1990 | 23,678 |
| *1980 | 25,905 |
| *1970 | 31,161 |
Due largely to the US EPAs Acid Rain Program, the U.S. has witnessed a 33 percent decrease in emissions between 1983 and 2002. This improvement resulted from flue gas desulfurization, a technology that enables SO2 to be chemically bound in power plants burning sulfur-containing coal or oil. In particular, calcium oxide reacts with sulfur dioxide to form calcium sulfite:
- CaO + SO2 → CaSO3
Aerobic oxidation converts this CaSO3 into CaSO4, gypsum. Most gypsum sold in Europe comes from flue gas desulfurization.
As of 2006, China is the world's largest sulfur dioxide polluter, with 2005 emissions estimated to be 25.49 million tons. This amount represents a 27% increase since 2000, and is roughly comparable with U.S. emissions in 1980.[2]
Al-Mishraq, an Iraqi sulfur plant, was the site of a 2004 disaster resulting in the release of massive amounts of sulfur dioxide into the atmosphere.
External links
- United States Environmental Protection Agency Sulfur Dioxide page
- International Chemical Safety Card 0074
- IARC Monograph "Sulfur Dioxide and some Sulfites, Bisulfites and Metabisulfites"
- NIOSH Pocket Guide to Chemical Hazards
- Food Intolerance Network - Sulfite factsheet
- Sulfur Dioxide, Molecule of the Month
Appendix: temperature dependence of aqueous solubility
22 g/100ml (0 °C) 15 g/100ml (10 °C) 11 g/100ml (20 °C) 9.4 g/100 ml (25 °C) 8 g/100ml (30 °C) 6.5 g/100ml (40 °C) 5 g/100ml (50 °C) 4 g/100ml (60 °C) 3.5 g/100ml (70 °C) 3.4 g/100ml (80 °C) 3.5 g/100ml (90 °C) 3.7 g/100ml (100 °C)




 216.73.216.110
216.73.216.110 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xxx
216.73.xxx.xxx
 Server Time:
Server Time:
