
Anthocyanins (from Greek: ἀνθός (anthos) = flower + κυανός (kyanos) = blue) are water-soluble vacuolar flavonoid pigments that appear red to blue, according to pH. They are synthesized exclusively by organisms of the plant kingdom, and have been observed to occur in all tissues of higher plants, providing color in leaves, stems, roots, flowers, and fruits.
Contents |
Function
In flowers, anthocyanin pigments function as pollinator attractants, and in fruits, the colorful skins attract animals which will eat the fruits and disperse the seeds. In photosynthetic tissues (such as leaves), anthocyanins have been shown to act as a "sunscreen", protecting cells from photo-damage by absorbing UV and blue-green light, thereby protecting the tissues from photoinhibition, or high light stress. This has been shown to occur in red juvenile leaves, autumn leaves, and broad-leaved evergreen leaves that turn red during the winter. It is also thought that red coloration of leaves may camouflage leaves from herbivores blind to red wavelengths, or signal unpalatability to herbivores, since anthocyanin synthesis often coincides with synthesis of unpalatable phenolic compounds.
In addition to their role as light-attenuators, anthocyanins also act as powerful antioxidants, helping to protect the plant from radicals formed by UV light and during metabolic processes. This antioxidant property is conserved even after consumption by another organism, which is another reason why fruits and vegetables with red skins and tissues are a nutritious food source.
Occurrence
| foodstuff | Anthocyanin in mg per 100 g foodstuff |
|---|---|
| blackcurrant | 190-270 |
| chokeberry | 200-1000 |
| aubergine | 750 |
| orange | ~200 |
| blackberry | ~115 |
| vaccinium | 80-420 |
| raspberry | 10-60 |
| cherry | 350-400 |
| redcurrant | 80-420 |
| red grape | 30-750 |
| red wine | 24-35 |
Not all land plants contain anthocyanin and in the Caryophyllales, Cactus and Galium mollugo they are replaced by betacyanins.
Anatomically, anthocyanins are found mostly in flowers and fruits but also in leaves, stems, and roots. In these parts they are found predominantly in outer cell layers such as the epidermis and peripheral mesophyll cells. The amounts are relatively large: one kilogram of blackberry for example contains approximately 1.15 gram, and red and black legumes can contain 20 gram per 1 kg. Other plants rich in anthocyanins are blackcurrant, chokeberry, cherry, eggplant, blue grape, Vaccinium and red cabbage and also the Usambara-violet. Anthocyanins are less abundant in banana, asparagus, pea, fennel, pear and potato. Most frequent in nature are the glycosides of cyanidin, delphinidin, malvidin, pelargonidin, peonidin and petunidin. Roughly 2% of all hydrocarbons fixated in photosynthesis are converted into flavonoids and their derivatives such as the anthocyanins. This is no less than 109 tons per year.

In plants anthocyanins are present together with other natural pigments like the closely chemically related flavonoids, carotenoids, anthoxanthins and betacyanins.
In still relatively young plants or new growth, where chlorophyll and wax production has not yet begun and which would be unprotected from UV light, anthocyanin production increases. Parts or even the whole plant are colored by these "juvenile anthocyanins," and thereby protected from damage. As soon as chlorophyll production begins, the production of the anthocyanin dye is reduced. The build-up of anthocyanin in plants is specific to the plant type, since it depends on the soil conditions, light, warmth and plant type and/or sort. Plants that have only a single anthocyanin as pigment is extremely rare, but occurs nevertheless. The absence or particularly strong prevalence for a certain anthocyanin in a plant is due to genetic circumstances.
Structure
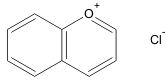
The pigment components of the anthocyanidins, the sugar-free anthocyanins, can be identified based on the structure of a large group of polymethine dye, as salt derivative of benzopyrylium (a cationic form of benzopyran, i.e an oxygen-containing heterocycle pyran fused to a benzene ring). The pyran ring in anthocyanin is connected to a phenyl group at the 2-position, which can carry different substituents. The counterion for the cationic oxygen in the pyran ring is mostly chloride. With this positive charge the anthocyanins differ from other flavonoids. The anthocyanins, anthocyanidins with sugar group, are mostly 3-glucosides of the anthocyanidins. In red wine, especially after maturation, many derivatives of the anthocyanins can be found.
The anthocyanins are subdivided into the sugar-free anthocyanidine aglycons and the anthocyanin glycosides. They are considered secondary metabolites and allowed as a food additive with E number 163. As of 2003 more than 400 anthocyanins had been reported[1] while more recent literature (early 2006), puts the number at more than 550 different anthocyanins. The difference in chemical structure that occurs in response to changes in pH is the reason why anthocyanins are often used as pH indicator, as they change from red in acids to blue in bases.

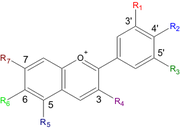
| Anthocyanidin | R1 | R2 | R3 | R4 | R5 | R6 | R7 |
| Aurantinidin | -H | -OH | -H | -OH | -OH | -OH | -OH |
| Cyanidin | -OH | -OH | -H | -OH | -OH | -H | -OH |
| Delphinidin | -OH | -OH | -OH | -OH | -OH | -H | -OH |
| Europinidin | -OCH3 | -OH | -OH | -OH | -OCH3 | -H | -OH |
| Luteolinidin | -OH | -OH | -H | -H | -OH | -H | -OH |
| Pelargonidin | -H | -OH | -H | -OH | -OH | -H | -OH |
| Malvidin | -OCH3 | -OH | -OCH3 | -OH | -OH | -H | -OH |
| Peonidin | -OCH3 | -OH | -H | -OH | -OH | -H | -OH |
| Petunidin | -OH | -OH | -OCH3 | -OH | -OH | -H | -OH |
| Rosinidin | -OCH3 | -OH | -H | -OH | -OH | -H | -OCH3 |
Biosynthesis
Anthocyanin pigments are assembled from two different streams of chemical raw materials in the cell: both starting from the C2 unit acetate (or acetic acid) derived from photosynthesis, one stream involves the shikimic acid pathway to produce the amino acid phenylalanine. The other stream (the acetic acid pathway) produces 3 molecules of malonyl-Coenzyme A, a C3 unit. These streams meet and are coupled together by the enzyme chalcone synthase (CHS), which forms an intermediate chalcone via a polyketide folding mechanism that is commonly found in plants. The chalcone is subsequently isomerized by the enzyme chalcone isomerase (CHI) to the prototype pigment naringenin, which is subsequently oxidized by enzymes such as flavanone hydroxylase (FHT or F3H), flavonoid 3' hydroxylase and flavonoid 3' 5'-hydroxylase. These oxidation products are further reduced by the enzyme dihydroflavonol 4-reductase (DFR) to the corresponding leucoanthocyanidins. It was believed that leucoanthocyanidins are the immediate precursors of the next enzyme, a dioxygenase referred to as anthocyanidin synthase (ANS) or leucoanthocyanidin dioxygenase (LDOX). It was recently shown however that flavan-3-ols, the products of leucoanthocyanidin reductase (LAR), are the true substrates of ANS/LDOX. The resulting, unstable anthocyanidins are further coupled to sugar molecules by enzymes like UDP-3-O-glucosyl transferase to yield the final relatively stable anthocyanins. More than five enzymes are thus required to synthesize these pigments, each working in concert. Any even minor disruption in any of the mechanism of these enzymes by either genetic or environmental factors would halt anthocyanin production.
Autumn Leaf Color
Many science text books incorrectly state that all autumn coloration (including red) is simply the result of breakdown of green chlorophyll, which unmasks the already-present orange, yellow, and red pigments (carotenoids, xanthophylls, and anthocyanins, respectively). While this is indeed the case for the carotenoids and xanthophylls (orange and yellow pigments), anthocyanins are not present until the leaf begins breaking down the chlorophyll, during which time the plant begins to synthesize the anthocyanin, presumably for photoprotection during nitrogen translocation.
Recent research
In December 2004 a peer-reviewed study at Michigan State University published by the American Chemical Society noted that anthocyanin could boost insulin production by up to 50%. However the study leader noted that despite the initial excitement, more study would be needed. Also in 2005, an article published in Applied and Environmental Microbiology demonstrated for the first time the biosynthesis of anthocyanins in bacteria [1].
References
- ^ http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=pubmed&dopt=Abstract&list_uids=14561507&query_hl=4&itool=pubmed_DocSum
- Andersen, O.M. Flavonoids: Chemistry, Biochemistry and Applications. CRC Press, Boca Raton FL 2006.
External links
- Anthocyanin Biosynthesis
- Red leaves - Catalyst ABC
- Chemicals Found in Cherries May Help Fight Diabetes
- Biochemicals found in dark raspberries may help fight Diabetes and Cancer (in German)
- A discussion of the role of anthocyanins in hydrangea coloration
- Anthocyanins FAQ MadSci Network Functions and uses as pH indicators or for pigment chromatography.




 216.73.216.133
216.73.216.133 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xxx
216.73.xxx.xxx
 Server Time:
Server Time: