 |
|
|
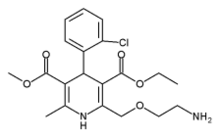
Amlodipine
|
|
| Systematic (IUPAC) name | |
|
3-ethyl-5-methyl-2-(2-aminoethoxymethyl)-4-(2- chlorophenyl)-1,4-dihydro-6-methyl-3,5- pyridinedicarboxylate benzenesulfonate |
|
| Identifiers | |
| CAS number | 88150-42-9 |
| ATC code | C08CA01 |
| PubChem | 2162 |
| DrugBank | APRD00520 |
| Chemical data | |
| Formula | C20H25ClN2O5.C6H6O3S |
| Mol. weight | 408.879 g/mol |
| Pharmacokinetic data | |
| Bioavailability | 64 to 90% |
| Metabolism | Hepatic |
| Half life | 30 to 50 hours |
| Excretion | Renal |
| Therapeutic considerations | |
| Pregnancy cat. | C(AU) C(US) |
| Legal status | POM(UK) ℞-only(US) |
| Routes | Oral (tablets) |
Amlodipine (as besylate, mesylate or maleate) is a long-acting calcium channel blocker used as an anti-hypertensive and in the treatment of angina. Amlodipine is marketed as Norvasc® in North America and as Istin® in the United Kingdom as well as under various other names. As other calcium channel blockers, amlodipine acts by relaxing the smooth muscle in the arterial wall, decreasing peripheral resistance and hence improving blood pressure; in angina it improves blood flow to the myocardium. It was developed under the direction of Dr. Simon Campbell.
Contents |
Indications
- hypertension
prophylaxis of angina
Cautions
- hepatic impairment
pregnancy
Contra-indications
- cardiogenic shock
unstable angina
significant aortic stenosis
breast feeding
Side effects
Some side effects of the use of amlodipine may be:
- Very often: peripheral edema (feet and ankles) - in
1 of 10 users
Often: dizziness, palpitations, muscle, stomach or headache, dyspepsia, nausea - in 1 in 100 users
Sometimes: blood disorders, development of breasts in men (gynecomastia), impotence, depression, insomnia, tachycardia - in 1 in 1,000 users
Rarely: erratic behavior, hepatitis, jaundice - in 1 in 10,000 users
Very rarely: hyperglycemia, tremor, Stevens-Johnson syndrome - in 1 in 100,000 users
Source: Sandoz product information sheet
Dose
- Hypertension or angina: 5 or 10 mg once daily.
Salts
In the United Kingdom tablets of amlodipine from different suppliers may contain different salts. The strength of the tablets is expressed in terms of amlodipine base. i.e. without the salt. Tablets containing different salts are therefore considered interchangeable.
Patent Loss
Pfizer patent protection on Norvasc until 2007. [1]
External links
- Norvasc - Prescription Drug Info Amlodipine Profile
- Istin - Summary of Product Characteristics from the electronic Medicines Compendium




 216.73.216.110
216.73.216.110 User Stats:
User Stats:
 Today: 0
Today: 0 Yesterday: 0
Yesterday: 0 This Month: 0
This Month: 0 This Year: 0
This Year: 0 Total Users: 117
Total Users: 117 New Members:
New Members:
 216.73.xxx.xxx
216.73.xxx.xxx
 Server Time:
Server Time:
